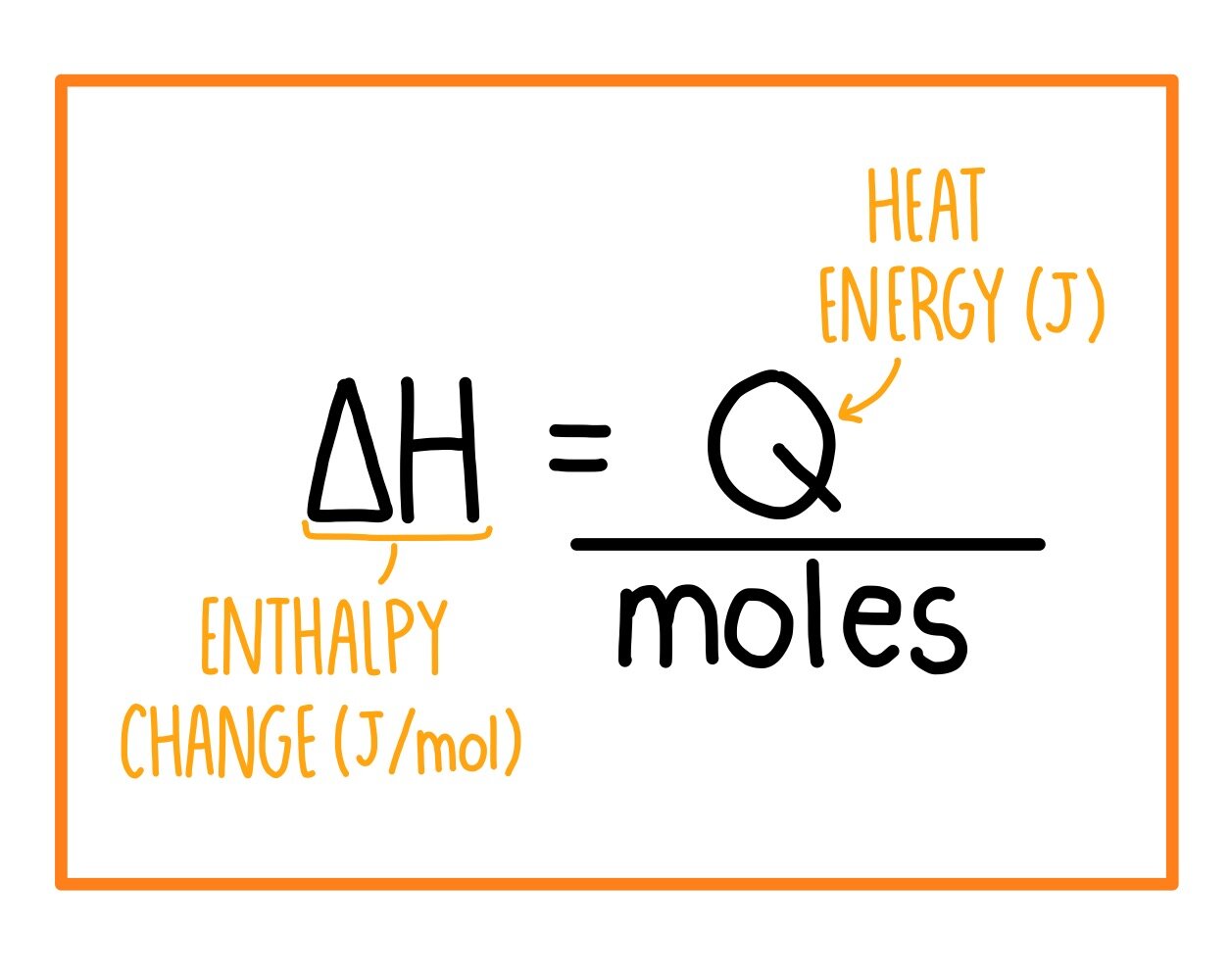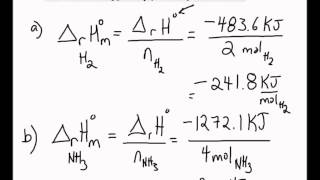Calculate the change in molar entropy and change in Gibbs' free energy when 1 mol of liquid butanol vaporizes at 25ºC to a gas that is at 0.001 atm? | Socratic
Experiment: To measure the molar enthalpy change for the dissolution ( enthalpy of solution) for vari - Tutorke

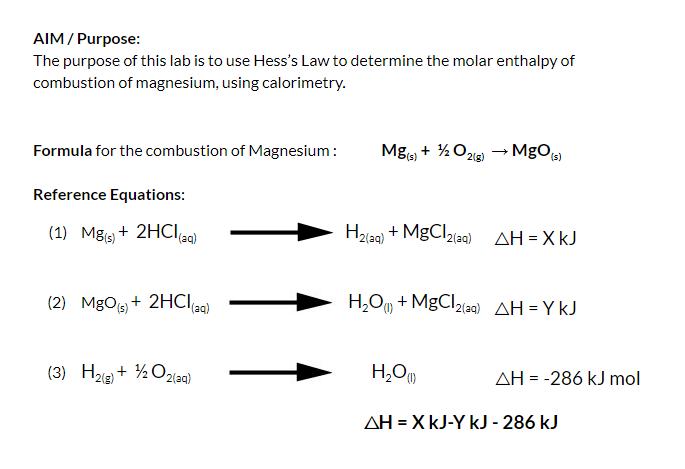
Molar Enthalpies. use proper scientific terminology to describe molar enthalpies calculate molar enthalpies Calculate molar enthalpies using the. - ppt download